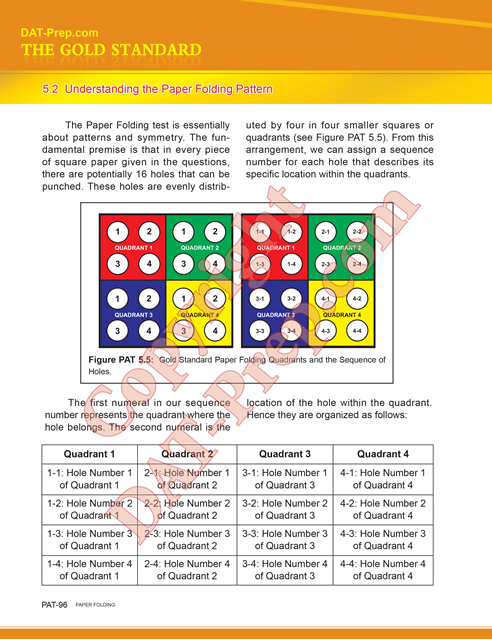
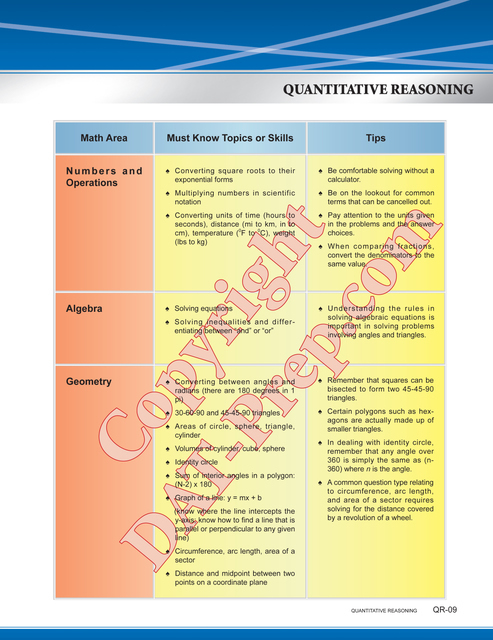
Purchase any Masters Series paperback book(s) and you will receive FREE ONLINE ACCESS to the eBook(s) while you await shipment. Start preparing TODAY!
Gold Standard DAT Organic Chemistry Review
(Dental Admission Test)
Gold Standard Organic Chemistry Book • Gold Standard DAT Organic Chemistry Table of Contents • DAT Organic Chemistry Test Content
- Title: DAT Organic Chemistry Masters Series, Review, Preparation and Practice for the Dental Admission Test by Gold Standard DAT
- SRP: $49.95
- Format: Paperback book or eBook
- Pages: 440 pages
- Edition: 2
- ISBN: 978-1-927338-63-6
- Publisher: RuveneCo Inc.
- Authors: DAT-prep.com, Gold Standard DAT
- Series: Book 3 of 6
- Description: Comprehensive chemistry review in color from the atom to thermodynamics to reaction mechanisms to NMR; General Chemistry Equation List & Organic Reaction Summary. Chapters begin with learning objectives and finish with practice multiple choice questions followed by useful explanations. This book also contains the content covering OAT Organic Chemistry.
- Paperback book
- PRICE: $44.95
$49.95
Temporary eBook pdf access available upon email request while awaiting shipment.
- Online eBook access for 6 months
- PRICE: $24.95
The eBook pdf is 100% online with the identical content as the paperback book.
Can't I use MCAT books instead?
There are many important DAT General and Organic Chemistry topics that are either not on the MCAT or are so rare as MCAT questions that the topics are not properly covered in MCAT books. Here is a partial list: general chemistry laboratory techniques and glassware, alkene, alkyne, ether and aromatic chemistry, multi-step organic syntheses, etc. The Gold Standard DAT was designed specifically for the DAT so all ADA topics are presented in color followed by chapter review questions, answers and explanations.
Table of Contents for Organic Chemistry
- Molecular Structure of Organic Compounds
- Stereochemistry
- Alkanes
- Alkenes
- Aromatics
- Alcohols
- Aldehydes and Ketones
- Carboxylic Acids
- Carboxylic Acids Derivatives
- Ethers and Phenols
- Amines
- Biological Molecules
- Separations and Purifications
- Spectroscopy
Key Organic Chemistry Reaction Mechanisms
Official ADA DAT Organic Chemistry Test Content (30 questions)
-
Mechanisms:
Energetics, and Structure - elimination, addition, free radical, substitution mechanisms, and other mechanisms and reactions.
-
Chemical and Physical Properties of Molecules:
Spectroscopy (1H NMR, 13C NMR, infrared, and multi-spectra), structure (polarity, intermolecular forces (solubility, melting/ boiling point, etc.), and laboratory theory and techniques (TLC, separations, etc.)
-
Stereochemistry (structure evaluation):
Chirality, isomer relationships, and conformations
-
Nomenclature:
IUPAC rules and functional groups in molecules
-
Individual Reactions of the Major Functional Groups and Combinations of Reactions to Synthesize Compounds:
Fertilization, descriptive embryology, developmental mechanisms.
-
Genetics:
Alkene/alkyne, aromatic, substitution/elimination, aldehyde/ketone, carboxylic acids and derivatives, and other. For each area listed above, the following sub-areas apply: general, one-step, and multi-step.
-
Acid-Base Chemistry:
Ranking acidity/ basicity (structure analysis and pH/ pKa data analysis), and prediction of products and equilibria
-
Aromatics and Bonding:
Concept of aromaticity, resonance, atomic/ molecular orbitals, hybridization, and bond angles/lengths.
In addition to the Dental Admission Test guides and software that we recommend to you, please consider visiting the official website for the Dental Admission Test: click on DAT in the US or click here: DAT for students in Canada.
 ×
×
FREE SHIPPING
Free shipping will be applied to addresses within the contiguous US with an estimated time for delivery of 2 to 10 days, the average being 6 days.
SHIPPING ADDRESS
We ship to the address associated with your Paypal account unless you specify otherwise at the time of payment.
SHIPPING TIME
We ship, on average, within the next 2 business days.
SHIPPING COSTS
Outside of contiguous US:
Shipping cost to Alaska/Hawaii/Puerto Rico/APO/FPO/DPO (US Military) is $6 by tracked USPS Media but it may take as long as 14 days for delivery.
Shipping and handling to address in Canada is $40 (tracked) with delivery within 7 days.
Shipping to an address outside of the US and Canada is calculated based on the destination and weight.
Email shipping@dat-prep.com with the destination and number of books for pricing.